Draw The Lewis Structure For The Xenon Difluoride Molecule
Draw The Lewis Structure For The Xenon Difluoride Molecule - If there are any atoms with a nonzero formal charge, be sure to write the formal charge next to the symbol. See an example of a molecule that violates the octet rule (xef₂) and learn how to draw. For the xef2 structure use the periodic table to find the total number of valence electrons for. Use vector addition to explain your answer. (2) draw single bonds between bonded atoms. Web lewis structure of xef2 contains two single bonds between the xenon (xe) atom and each fluorine (f) atom. The xenon atom (xe) is at the center and it is surrounded by 2 fluorine atoms (f). (4 points)what is the molecular shape of xenon difluoride? Xenon has eight valence electrons, and each fluorine atom has seven valence electrons, so the total number of valence electrons is 22. Determine the total number of valence electrons in xef2 by adding the valence electrons of each atom. Web i quickly take you through how to draw the lewis structure of xef2 (xenon difluoride). A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. (5 points) draw the lewis structure for xenon difluoride. See an example of a molecule that violates the octet rule (xef₂) and learn how to draw. Figure out. Use vector addition to explain your answer. Web draw the lewis structure for the xenon difluoride (xef2) molecule. Let’s draw and understand this lewis dot structure step. Xef2 lewis structure involves 1 atom of xenon and 2 atoms of fluorine. Web xef2 lewis structure + molecular geometry chem101csub 3.77k subscribers subscribe 43 14k views 9 years ago chemistry learning made. See an example of a molecule that violates the octet rule (xef₂) and learn how to draw its lewis diagram in this video. Web in xef2 molecule, two fluorine atoms are arranged symmetrically on the outside with the central atom xenon in the middle. Find the total valence electrons in xef2 molecule in order to find the total valence electrons. Web what is the lewis structure of xef2? Web xenon difluoride formula : Web the xef6 lewis structure refers to the arrangement of atoms and electrons in a molecule of xenon hexafluoride.xenon hexafluoride is a compound composed of one xenon atom bonded to six fluorine atoms. Web draw the lewis structure for the xenon difluoride (xef2) molecule. Lewis diagram of. When constructing a lewis diagram, keep in mind the octet rule, which refers to the tendency. Web i quickly take you through how to draw the lewis structure of xef2 (xenon difluoride). Hence xenon difluoride is nonpolar as there is no polarity observed in the molecule. Web xef2 lewis structure is the abbreviation of xenon difluoride. (1) find the number. It is one of those rare compounds which involve noble gases despite their strong stability. The lewis structure of xef2 depicts the arrangement of atoms and valence electrons in a molecule of xenon difluoride. It shows xenon (xe) as the central atom bonded to two fluorine (f) atoms. (b) (2 points) c the electronegativity of xe is 2.6. (4 points)what. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Find the total valence electrons in xef2 molecule in order to find the total valence electrons in xef2 (xenon difluoride) molecule, first of all you should know the valence electrons present in xenon atom as well as fluorine atom. Each fluorine atom forms a. See an example of a molecule that violates the octet rule (xef₂) and learn how to draw its lewis diagram in this video. Web the lewis diagram of a molecule can be constructed using the following stepwise procedure: This problem has been solved! Each fluorine atom forms a single bond with the. Trigonal bipyramidal molecular geometry :. Perspective drawing with bond angles: Xef2 lewis structure involves 1 atom of xenon and 2 atoms of fluorine. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web a video explanation of how to draw the lewis dot structure for xenon difluoride, along with information about the compound including formal charges, polarity, hybrid. Xenon has eight valence electrons, and each fluorine atom has seven valence electrons, so the total number of valence electrons is 22. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Let’s draw and understand this lewis dot structure step. Web the xef6 lewis structure refers to the arrangement of atoms and electrons. However, in the excited state, its configuration will change to 5s 2 5p 5 5d 1. Web xenon difluoride formula : The lewis structure of xef6 shows that xenon is the central atom, surrounded by six fluorine atoms. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. (2) draw single bonds between bonded atoms. Web in xef2 molecule, two fluorine atoms are arranged symmetrically on the outside with the central atom xenon in the middle. Web watch on steps of drawing xef2 lewis structure step 1: The xenon atom (xe) is at the center and it is surrounded by 2 fluorine atoms (f). (4 points)what is the molecular shape of xenon difluoride? Hence xenon difluoride is nonpolar as there is no polarity observed in the molecule. Web the structure of xenon difluoride is illustrated below. The lewis structure of xef2 depicts the arrangement of atoms and valence electrons in a molecule of xenon difluoride. Web the xef6 lewis structure refers to the arrangement of atoms and electrons in a molecule of xenon hexafluoride.xenon hexafluoride is a compound composed of one xenon atom bonded to six fluorine atoms. Web the lewis diagram of a molecule can be constructed using the following stepwise procedure: Xef2 lewis structure involves 1 atom of xenon and 2 atoms of fluorine. The electronegativity of f is 4.0.
Hello Guys! Today we are going to look at the Lewis Structure of XeF2
xenon difluoride Overview, Structure, Properties & Uses

XeF2 Lewis Structure How to Draw the Lewis Structure for XeF2 YouTube

XeF2 Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram

The Lewis Structure Of Xef2 Understanding The Bonding Of Xenon

Xenon difluoride, 99.5+, Thermo Scientific Chemicals Fisher Scientific
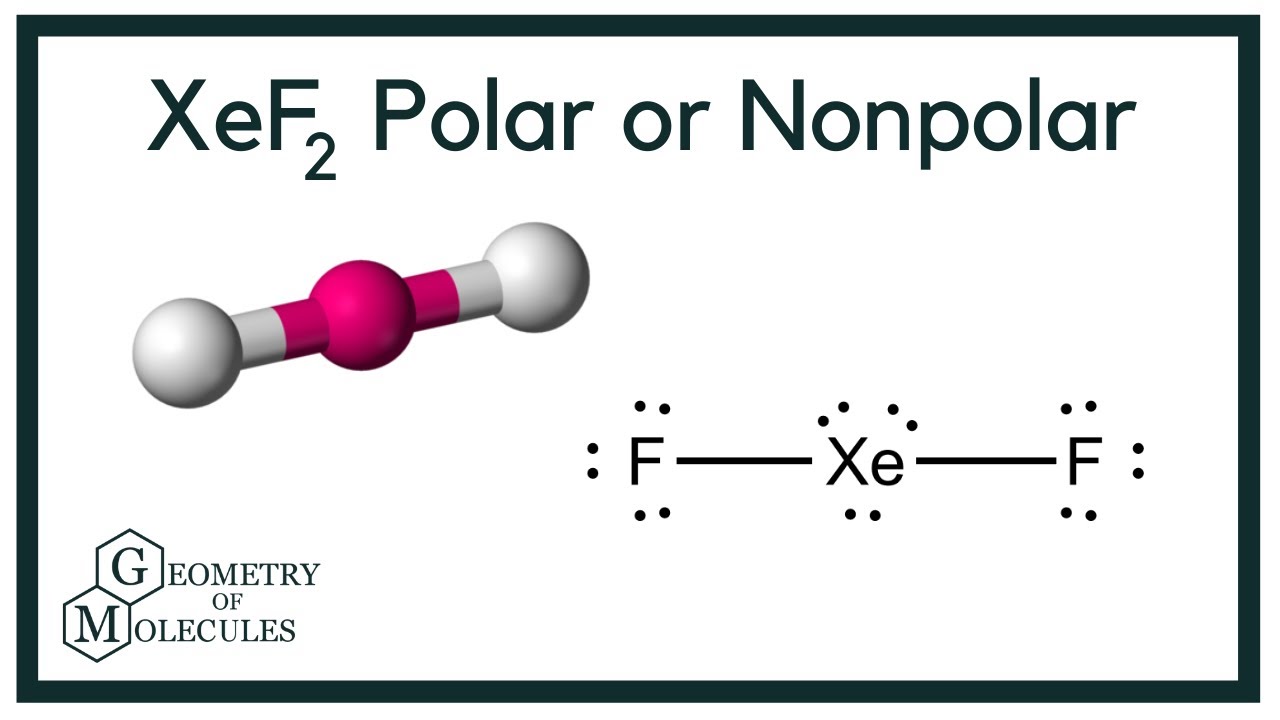
Xenon Difluoride Shape Draw Easy

Xenon Difluoride Molecular Structure Isolated on Black. 3d Illustration

Number of Lone Pairs and Bonding Pairs for XeF2 (Xenon difluoride

XeO2F2 Lewis Structure How to Draw The Lewis Structure for XeO2F2
(3) Distribute The Remaining Electrons Throughout The Molecule, Keeping In Mind The Duet And Octet Rules.
(B) (2 Points) C The Electronegativity Of Xe Is 2.6.
For The Xef2 Structure Use The Periodic Table To Find The Total Number Of Valence Electrons For.
See An Example Of A Molecule That Violates The Octet Rule (Xef₂) And Learn How To Draw.
Related Post: